- Blog
- Microsoft office 365 excel download free
- Christmas light projector
- Free professional resume templates download
- Periodic table is molar mass or amu
- Izuhara map ghost of tsushima liberation
- Bmi calculator chart women
- Downloader youtube app for pc windows 7
- Play vietcong windows 10
- TechSmith Camtasia-s New Asset Library
- Free movie maker download windows 7
- Bingo number generator caller
- Soulcalibur iv torrent
- System mechanic pro 18 torrent download crack
- Afterglow ps3 pc controller menu
- Disable mac function keys xplane 11
- Multiplication 14 times table worksheets
- 4500k hid vs halogen
- Keto meal planner based on macros
- Excel household budget worksheet
- Villanova charles barkley daughter
- Ancient warrior glyph
- White youtube banner
- Vocaloid vsq vpr
- Visual novel reader download 2020
- Pdf editor online download free
- Html iframe code not displaying
- Number of valence electrons periodic table
- Printable calendar budget planner
- Geometry dash game play for free
- Type o negative blood diet
- Sims 4 kids cc
- Artistic Licence Holdings
- Dragon age inquisition elven glyphs ancient baths
- Color blind test for kids blue green
- Wolf among us logo png
- Wedding emcee script
- The coaching tools company wheel of life
- Guitar hero 3 song pack clone hero
- Waifu sex simulator importing models
- Adobe movie maker free download
As a result of this standard, the mass of all other elements on the periodic table are determined relative to carbon-12. So, the mass of one unit of #"NaCl"# is #58.44#"amu"#, and the mass of one mole of #"NaCl"# is #58.44#"grams"#. This is the standard unit for atomic or molecular mass, and 1 amu is thus 1/12 th the mass of a 12 C atom. We know that the mass of one mole of carbon-12 atoms is 12 'g', or, 1 'mol'm(''12C)12 'g', because of the definition of the mole. We now need to convert from 'amu' to 'g/mol'. The periodic table lists the atomic mass of carbon as 12. This is numerically equivalent to the molar mass of #"NaCl"#. On the periodic table, it shows us that calcium has a mass of around 40 'amu'. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. The atomic mass of #"NaCl"# can be found by using the atomic masses of the individual elements found on a periodic table: To illustrate this fact, let's look at the compound sodium chloride ( #"NaCl"#). The atomic mass in #"amu"# of a substance is numerically equivalent to the mass in #"g"# of one mole of that substance. Thus, since the atomic mass of iron is 55.847 amu, one mole of iron atoms would weigh 55.847 grams. The molar mass is an average of many instances of the compound, which often vary in mass. 1 The molar mass is a bulk, not molecular, property of a substance. So finally we get that 1 g/mol 1 amu 1 g / m o l 1 a m u. The atomic mass is useful in chemistry when it is paired with the mole concept: the atomic mass of an element, measured in amu, is the same as the mass in grams of one mole of an element. In chemistry, the molar mass ( M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. molecular mass of H 2 O (2 x 1.008 amu) + (15.999 amu) molecular mass of H 2 O 2.016 amu + 15. The molecular mass of water will be 2 masses of hydrogen plus the mass of oxygen.
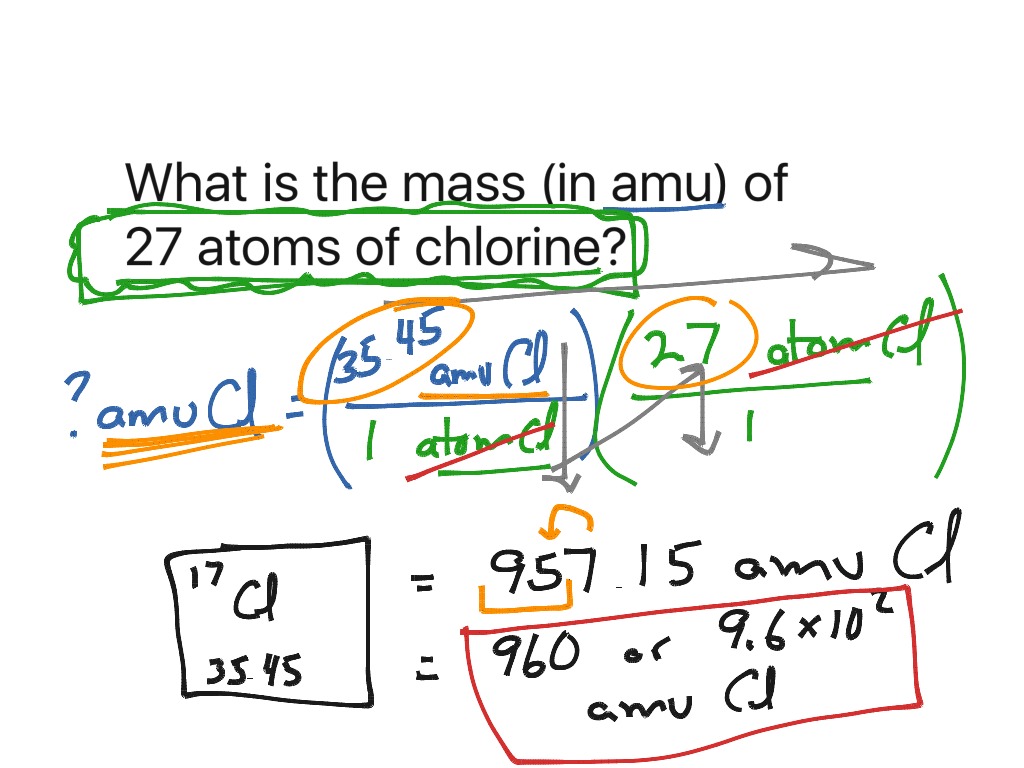
Molar mass is the mass of one mole of a substance.Ītomic mass is the mass of one individual unit of a substance.Īn important concept regarding the mole is that m(X12X2122C) 12 amu 12 g/mol m ( X 12 X 2 2 12 C) 12 a m u 12 g / m o l. The periodic table shows the atomic mass of hydrogen is 1.0008 amu and the atomic mass of oxygen is 15.999 amu.
- Blog
- Microsoft office 365 excel download free
- Christmas light projector
- Free professional resume templates download
- Periodic table is molar mass or amu
- Izuhara map ghost of tsushima liberation
- Bmi calculator chart women
- Downloader youtube app for pc windows 7
- Play vietcong windows 10
- TechSmith Camtasia-s New Asset Library
- Free movie maker download windows 7
- Bingo number generator caller
- Soulcalibur iv torrent
- System mechanic pro 18 torrent download crack
- Afterglow ps3 pc controller menu
- Disable mac function keys xplane 11
- Multiplication 14 times table worksheets
- 4500k hid vs halogen
- Keto meal planner based on macros
- Excel household budget worksheet
- Villanova charles barkley daughter
- Ancient warrior glyph
- White youtube banner
- Vocaloid vsq vpr
- Visual novel reader download 2020
- Pdf editor online download free
- Html iframe code not displaying
- Number of valence electrons periodic table
- Printable calendar budget planner
- Geometry dash game play for free
- Type o negative blood diet
- Sims 4 kids cc
- Artistic Licence Holdings
- Dragon age inquisition elven glyphs ancient baths
- Color blind test for kids blue green
- Wolf among us logo png
- Wedding emcee script
- The coaching tools company wheel of life
- Guitar hero 3 song pack clone hero
- Waifu sex simulator importing models
- Adobe movie maker free download
